Periodic Table
A table of the chemical elements discovered that is arranged in order of atomic number, usually in rows so that the elements with similar atomic structure (similar chemical properties) appear in vertical columns. The Russian chemist Dmitri Mendeleev was the first scientist to make a periodic table similar to the one used today. Mendeleev arranged the elements by atomic mass, corresponding to relative molar mass.
|
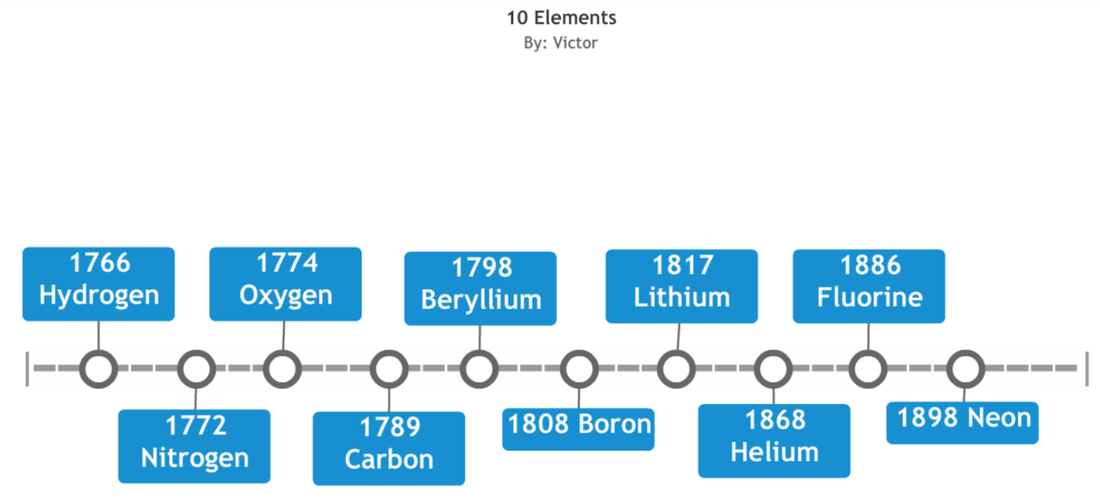
(H) 1766 Hydrogen – Henry Cavendish was the first to recognize
hydrogen gas as a discrete substance hence naming the gas in Greek from a
metal-acid reaction “flammable air”. Robert was the founder of Hydrogen gas in
1671 by describing the reaction between iron fillings and dilute acids which
results the production of hydrogen gas.
(He) 1868 Helium – A French astronomer Jules Janssen and Norman Lockyer detected an unknown yellow spectral line signature in sunlight during a solar eclipse. He named it Helium meaning in Greek, God of the sun Helios. (Li) 1817 Lithium – Johann August Arfvedson was the discoverer of Lithium in the mineral petalite (LiAl(Si2O5)2) however was isolated by William Thomas Brande and Sir Humphrey Davy through the electrolysis of Lithium oxide (Li2O). It is a Greek word “Lithos” meaning “stone”. (Be) 1798 Beryllium – A French chemist Louis-Nicolas Vauquelin suggested the name glucinium meaning “sweet tasting,” for the element because the element and some of its compounds have a sweet taste. (B) 1808 Boron – Joseph-Louis Gay-Lussac and Louis-Jaques Thenard and independently by Sir Humphry Davy discovered Boron by combining boric acid (H3BO3) with potassium. Amorphous boron (meaning the atoms are bonded together in a random order) in the form of a brown powder. (C) 1789 Carbon – The discovery of Carbon was known since ancient times. It was first recognized as an element in the second half of the 18th century by Antonie Lavoisier. He proposed the name carbo meaning “charcoal or burnt wood” in the language of Latin and showed that rare crystals are made up carbon. |
(N) 1772 Nitrogen – Daniel Rutherford discovered Nitrogen by removing oxygen and carbon dioxide from air and showed that the residual gas would not support combustion or living organisms. It was named noxious air but Scheele, Cavendish, Priestley, and others at about the same time studied “Burnt” or “dephlogisticated” air, as air without oxygen was then called Nitrogen.
(O) 1774 Oxygen – Oxygen had been produced by several chemists prior to its discovery however they failed to recognize it as a distinct element. Joseph Priestley and Carl Wilhelm Scheele both independently discovered oxygen, but Priestly is usually given credit for the discovery. It was discovered by heating several compounds including potassium nitrate, manganese oxide and mercury oxide as the outcome was found as the gas was released which enhanced combustion. (F) 1886 Fluorine – Henri Moissan discovered Fluorine by collecting the gas by passing an electric current through one of its compounds, hydrogen fluoride (H2F2) and observing its reaction. Fluorine is the most active chemical element reacting with virtually every element. It was named “Fluere” meaning flow or flux. (Ne) 1898 Neon – Sir William Ramsay and Morris Travers discovered the element of krypton. Like krypton neon was discovered through the study of liquefied air. Neon was named “neo” meaning new. |
3 Scientists
Dmitri Mendeleev8th February 1834 - 2nd February 1907
Dmitri Ivanovich Mendeleev was a Russian chemist and an inventor. He created the Periodic Law, created another version of the periodic table of elements, and used it to correct the properties of some discovered elements and also predicted the properties of eight elements yet to be. The table consists of arrangements of their atomic number, weight, exhibit. The elements which are similar regarding their chemical properties have atomic weights which are either of nearly the same value. |
Daniel Rutherford
3rd November 1749 – 15th December 1819
Daniel Rutherford was a Scottish physician, chemist and botanist who is most famous for the isolation of Nitrogen. He found that carbon dioxide cannot burn a candle. He then tested out with a living mouse in a confined space. He burned a candle in the air and then phosphorus in it then the air passed through a carbon dioxide absorbing solution. The remaining oxygen did not support combustion and the mouse could not live. Daniel called the gas "Noxious air or "Phlogisticated air" |
Henry Cavendish10th October 1731 - 24th February 1910
Henry Cavendish was a British natural philosopher, scientist and an important experimental and theoretical chemist and physicist. Cavendish is noted for his discovery of Hydrogen or what he called "inflammable air". He described the density of inflammable air, which formed water on combustion, in a 1766 paper "On Factitious Airs". |
The Format of the Periodic Table
The numbers on the top left indicates the atom's Atomic number as it is ordered from top to bottom, left to right.
Mass number sometimes founder under each atom on the periodic table. Again it is listed in order from top to bottom, left to right.
Alkali metals are the chemical elements found in group 1. except Hydrogen (Li
Alkaline earth metals have their outermost electrons in an s-orbital. It is found in group 2 vertically.( Be, Mg, Ca, Sr, Ba, Ra)
Transitional Metals have advanced shells and orbit. They have a lot of electrons and distribute them in different ways. They're found in the middle section of group 3-12.
Metalloids are found along the step like lines between metals and non-metals of the periodic table. (B, Si, Ge, As, Sb, Te, Po)
Non-metals are found within the corners. Those non-metals are more stable than metals thus having to be on the side except Hydrogen.
The Halogen group is found in group 17
All of the elements in group 18 are noble gases. These elements don't like to react because it is stable unlike other atoms.They're found on the right side of the table.
Periods are groups horizontally in the periodic table. The atomic number increases from left to right. The periods are numbered 1 through 7 and on the left hand side of the table.
The groups are allocated within groups vertically. They represent the same number of electrons in the outer orbital.
Mass number sometimes founder under each atom on the periodic table. Again it is listed in order from top to bottom, left to right.
Alkali metals are the chemical elements found in group 1. except Hydrogen (Li
Alkaline earth metals have their outermost electrons in an s-orbital. It is found in group 2 vertically.( Be, Mg, Ca, Sr, Ba, Ra)
Transitional Metals have advanced shells and orbit. They have a lot of electrons and distribute them in different ways. They're found in the middle section of group 3-12.
Metalloids are found along the step like lines between metals and non-metals of the periodic table. (B, Si, Ge, As, Sb, Te, Po)
Non-metals are found within the corners. Those non-metals are more stable than metals thus having to be on the side except Hydrogen.
The Halogen group is found in group 17
All of the elements in group 18 are noble gases. These elements don't like to react because it is stable unlike other atoms.They're found on the right side of the table.
Periods are groups horizontally in the periodic table. The atomic number increases from left to right. The periods are numbered 1 through 7 and on the left hand side of the table.
The groups are allocated within groups vertically. They represent the same number of electrons in the outer orbital.